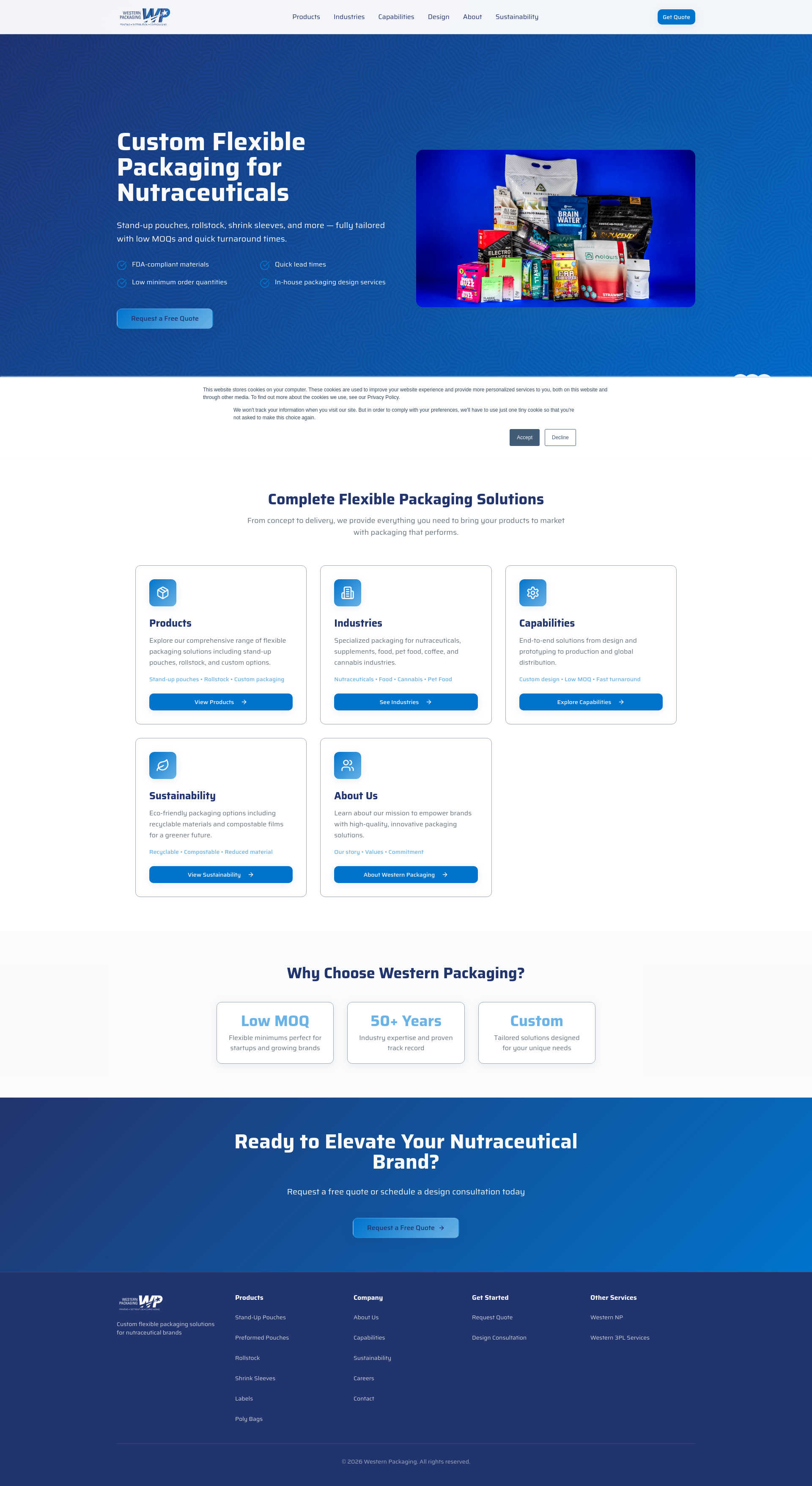
Discover how advanced flexible rollstock packaging is revolutionizing nutraceutical product protection, shelf appeal, and sustainability in the rapidly evolving health supplement market.
Introduction
Nutraceutical brands face a unique set of packaging challenges in 2026. Products like protein powders, pre-workout supplements, collagen, greens, and capsules demand packaging solutions that protect product quality, extend shelf life, run efficiently on automated equipment, and present the brand professionally at retail or direct-to-consumer touchpoints.
Flexible rollstock packaging has become the preferred solution for nutraceutical packaging buyers, supplement brands, engineers, and operations teams. It delivers high-barrier protection, custom printing capabilities, production efficiency, and format versatility—all while supporting the unique requirements of packaging for nutraceutical companies. At Western Packaging, we help companies share their products with the world by making flexible packaging truly flexible, adapting to your product specifications, equipment needs, and brand goals.
What Is Flexible Rollstock Packaging?
Flexible rollstock packaging refers to flexible packaging films supplied on rolls and designed for use on automated form-fill-seal (FFS) equipment. These rolls are loaded onto vertical form-fill-seal (VFFS) or horizontal form-fill-seal (HFFS) machines that form the film into a specific package shape, fill it with product, and seal it—all in a continuous, automated process.
Rollstock is used to create a wide range of flexible formats including stand-up pouches, flat pouches, sachets, stick packs, flow wraps, and pillow bags. Because the film is supplied on a roll rather than as pre-made bags, it integrates seamlessly with high-speed filling lines, reducing material handling and enabling efficient, repeatable production runs. This makes rollstock packaging suppliers critical partners for brands scaling nutraceutical production while maintaining consistent quality and appearance.
Why Nutraceutical Brands Are Transitioning to Flexible Rollstock Solutions
Nutraceutical products are sensitive to environmental factors like moisture, oxygen, light, and temperature. Flexible rollstock packaging addresses these concerns through engineered barrier properties that preserve product efficacy, flavor, texture, and shelf life. High-barrier flexible packaging films can be tailored to the specific requirements of each product, whether it's a hygroscopic powder, an oil-based softgel, or a moisture-sensitive tablet.
Custom printed rollstock supports brand differentiation in competitive retail environments. Brands can incorporate full-color graphics, gradients, matte or gloss finishes, and detailed product information directly on the film. This eliminates the need for secondary labeling in many cases and ensures a consistent, professional appearance across production runs.
Flexible rollstock also enables efficient production. Automated filling lines running rollstock achieve higher throughput, lower labor costs, and reduced material waste compared to manual or semi-automated processes. The ability to offer flexible sizes—from single-serving stick packs to bulk pouches—gives brands the versatility to serve different market segments, sampling programs, subscription models, and retail formats without retooling equipment or maintaining separate packaging inventories.
Compatibility with automated filling systems is another major advantage. Rollstock integrates with VFFS and HFFS machines used across nutraceutical facilities, supporting consistent fill weights, seal integrity, and minimizing downtime. This makes rollstock a practical choice for operations teams managing high-volume production schedules and quality standards.
Common Nutraceutical Products Packaged in Rollstock Formats
Flexible rollstock packaging serves a diverse range of nutraceutical products. Protein powders, pre-workout formulas, and greens powders are commonly packaged in stand-up pouches or pillow bags with resealable zippers for convenience and product protection. Collagen peptides, both flavored and unflavored, benefit from high-barrier rollstock that prevents moisture ingress and maintains powder flowability.
Capsules and tablets are often packed in flow wraps, sachets, or small pouches that protect against humidity and light exposure. Gummies, which are particularly sensitive to moisture and temperature, require rollstock films with strong moisture barriers and sealant layers that maintain package integrity throughout the product's shelf life.
Drink mixes, electrolyte powders, and single-serve supplements are ideal candidates for stick packs and sachets produced from rollstock. These formats offer portion control, portability, and ease of use—key factors for consumer convenience. Sample packs and trial sizes also leverage rollstock's flexibility, allowing brands to test new formulations or enter new distribution channels without significant packaging investment.
Essential Barrier Properties and Material Technologies for Supplement Protection
The performance of nutraceutical packaging depends on selecting the right combination of film materials and barrier layers. Common substrates include polyethylene terephthalate (PET), polyethylene (PE), oriented polypropylene (OPP), and metallized PET (MET PET). These materials are often combined in multi-layer laminations to achieve specific barrier and mechanical properties.
High-barrier laminations typically include aluminum foil or metallized layers that provide exceptional protection against moisture, oxygen, light, and aroma transfer. Foil structures are the gold standard for products with stringent shelf-life requirements, such as probiotics, enzymes, and botanicals sensitive to oxidation. Metallized films offer a balance between barrier performance and cost, making them suitable for products with moderate sensitivity.
Sealant layers, usually composed of polyethylene or other heat-sealable polymers, ensure strong, consistent seals that resist leaks, punctures, and contamination. Seal integrity is critical for maintaining product quality and preventing costly recalls or customer complaints. Advanced sealant technologies enable sealing through product contamination, a common challenge in powder filling operations.
Moisture barrier performance is measured by water vapor transmission rate (WVTR), while oxygen barrier is assessed through oxygen transmission rate (OTR). Nutraceutical packaging buyers should specify target WVTR and OTR values based on product sensitivity, expected shelf life, and storage conditions. Light barrier, provided by opaque or metallized films, protects photosensitive ingredients like vitamins and certain botanicals. Aroma barrier prevents flavor loss and protects against off-flavors caused by external odors.
Puncture resistance and mechanical strength ensure that packages withstand handling, shipping, and retail display without compromising product protection. Film gauge, tensile strength, and tear resistance are key specifications that impact both performance and production efficiency. Matte and gloss finishes offer aesthetic differentiation and can enhance shelf presence, with matte finishes often perceived as premium and gloss finishes providing vibrant color reproduction.
Recyclable and sustainable film options are increasingly important for brands responding to consumer demand and corporate sustainability goals. Mono-material structures, such as all-polyethylene laminates, improve recyclability compared to traditional multi-material films. While not all recyclable films are suitable for every application, rollstock packaging suppliers can help brands evaluate options that balance performance, cost, and environmental impact.
Sustainability Meets Performance in Modern Rollstock Film Structures
Sustainability is a defining consideration for nutraceutical packaging in 2026. Brands and consumers alike are pushing for packaging solutions that reduce environmental impact without sacrificing product protection or shelf appeal. Modern rollstock film structures are evolving to meet these demands through material innovation, lightweighting, and design for recyclability.
Mono-material film structures, composed of a single polymer family, are designed to be compatible with existing recycling streams. For example, all-PE laminates can often be recycled alongside other polyethylene films, reducing landfill waste and supporting circular economy initiatives. While these structures may not achieve the same barrier performance as traditional foil or multi-material laminates, they are suitable for many nutraceutical applications with moderate shelf-life requirements.
Lightweighting reduces the amount of material used per package, lowering carbon footprint, shipping costs, and raw material consumption. Advances in film extrusion and lamination technology enable thinner films that maintain mechanical strength and barrier performance. This approach benefits both sustainability goals and operational efficiency.
Recyclable options should be evaluated on a case-by-case basis. Not every nutraceutical product can be packaged in recyclable films without compromising quality, and brands must balance performance, cost, regulatory compliance, and environmental objectives. Rollstock packaging suppliers with expertise in sustainable film development can guide brands through material selection, testing, and supply chain integration.
It's also important to recognize that sustainability extends beyond material composition. Efficient production processes, reduced material waste, optimized package sizes, and supply chain logistics all contribute to a packaging solution's overall environmental impact. At Western Packaging, we help companies share their products with the world by making flexible packaging truly flexible, adapting to both product requirements and sustainability commitments.
Custom Printing Capabilities That Elevate Shelf Presence and Brand Identity
Custom printed rollstock is a powerful tool for nutraceutical brands competing for attention in crowded retail environments and online marketplaces. High-quality printing transforms functional packaging into a brand asset, communicating product benefits, ingredient transparency, and brand values directly to consumers.
Flexographic and rotogravure printing are the primary methods used for custom printed rollstock. Flexographic printing offers cost-effective, high-speed production suitable for medium to long runs, while rotogravure delivers superior image quality and color consistency for premium brands and complex graphics. Digital printing is emerging as an option for short runs, customization, and rapid prototyping, though it is not yet as widely adopted for high-volume nutraceutical production.
Printing supports essential brand and regulatory elements. Supplement Facts panels, ingredient lists, allergen warnings, UPC barcodes, lot codes, and expiration dates must be clearly legible and compliant with FDA and other regulatory requirements. Custom printed rollstock can accommodate these elements without compromising design aesthetics or requiring secondary labeling.
Consistent brand appearance across product lines and SKUs reinforces brand recognition and consumer trust. Custom printing ensures that colors, fonts, logos, and messaging remain uniform across production runs and packaging formats. This consistency is especially important for brands with multiple products, flavors, or sizes.
Advanced printing techniques enable special effects such as matte and gloss contrast, metallic inks, tactile coatings, and transparent windows. These features enhance shelf appeal and provide tactile and visual differentiation. Transparent windows, for example, allow consumers to see the product inside, which can be particularly effective for gummies, capsules, or brightly colored powders.
Artwork support from experienced rollstock packaging suppliers ensures that designs are print-ready, color-matched, and optimized for the selected printing process. This reduces delays, minimizes reprints, and helps brands bring new products to market faster.
Labeling and Compliance Considerations for Nutraceutical Rollstock Packaging
Nutraceutical packaging must accommodate extensive labeling and regulatory requirements. Flexible rollstock provides ample space for Supplement Facts panels, ingredient lists, allergen statements, net weight declarations, manufacturer information, and country of origin labeling. These elements are typically integrated directly into the custom printed rollstock design, ensuring clarity, legibility, and compliance.
Barcodes, including UPC and GS1-compliant codes, are essential for retail distribution and inventory management. Custom printed rollstock can incorporate barcodes in optimal locations that support scanning accuracy without interfering with design aesthetics.
Lot codes, date codes, and expiration dates are critical for traceability, quality control, and consumer safety. These variable data elements are typically applied using thermal transfer, inkjet, or laser coding systems during or after the filling process. Rollstock films should be compatible with these coding technologies to ensure legible, durable marks that withstand handling and storage.
Regulatory compliance in the nutraceutical industry is complex and varies by product type, ingredient claims, and distribution channels. Brands are responsible for ensuring that their packaging meets FDA regulations, state-level requirements, and industry-specific guidelines. Western Packaging does not provide legal or regulatory advice, and brands should consult with qualified regulatory professionals or legal counsel to ensure full compliance.
In addition to regulatory requirements, packaging should support clear communication of product benefits, usage instructions, and brand messaging. Well-designed labeling enhances consumer trust, reduces questions and returns, and strengthens brand loyalty.
Optimizing Supply Chain Efficiency Through Strategic Packaging Design
Production fit is a critical but often overlooked aspect of flexible rollstock packaging. Even the best-designed film will underperform if it's not compatible with your filling equipment, production line speed, and operational workflows. Engineers and operations teams must evaluate several technical specifications to ensure smooth, efficient production runs.
Film gauge, or thickness, affects mechanical strength, barrier performance, and machine compatibility. Thicker films offer better puncture resistance and barrier properties but may require adjustments to sealing temperature, dwell time, or machine settings. Thinner films reduce material costs and improve sustainability metrics but must still meet performance standards.
Web width and roll size must match your equipment specifications. VFFS and HFFS machines are designed for specific web widths, and using the wrong size can result in waste, downtime, or poor seal quality. Roll size, including diameter and core size, impacts how often rolls need to be changed and how much material can be stored on the production floor.
Core size, typically 3-inch or 6-inch, must be compatible with your machine's roll handling system. Using the correct core size reduces setup time and minimizes the risk of roll slippage or film damage during production.
Sealant compatibility is essential for achieving strong, consistent seals. The sealant layer must be compatible with your machine's sealing jaws, temperature range, and dwell time. Incompatible sealants can result in weak seals, leaks, or excessive waste.
Registration marks, also called eye marks, are printed alignment guides that ensure accurate printing, cutting, and sealing. Properly positioned registration marks enable your equipment to maintain consistent package dimensions and print placement, reducing waste and rework.
Coefficient of friction (COF) and slip properties affect how the film feeds through your equipment. Films with inappropriate COF can stick together, slip on rollers, or fail to advance correctly, leading to jams, misalignment, and downtime. Rollstock packaging suppliers should provide COF specifications and recommendations based on your equipment and production environment.
Machine compatibility extends beyond basic dimensions. Factors like film memory, stiffness, and heat resistance all influence how the film performs on your line. Testing new films on your equipment before committing to large orders is a best practice that prevents costly surprises.
Run speed and waste reduction are closely linked. Films optimized for your equipment and production parameters enable higher throughput, fewer stoppages, and less material waste. This translates to lower cost per unit, improved production scheduling, and better overall efficiency.
Downtime prevention starts with selecting the right film and supplier. Reliable rollstock packaging suppliers provide consistent quality, accurate specifications, technical support, and responsive service. These factors directly impact your ability to meet production targets and deliver products on time.
Choosing a Supplier: What Nutraceutical Packaging Buyers Should Ask
Selecting the right rollstock packaging supplier is as important as choosing the right film structure. Nutraceutical packaging buyers, engineers, and operations teams should evaluate suppliers based on technical capabilities, service quality, and long-term partnership potential.
Printing capabilities should be a top consideration. Ask about printing methods, color matching processes, proof approval workflows, and turnaround times. Suppliers with in-house printing capabilities can offer better control over quality, lead times, and design iterations.
Film structures and barrier options are critical for product protection. A knowledgeable supplier will ask detailed questions about your product, shelf-life requirements, storage conditions, and distribution channels. They should be able to recommend film constructions, explain trade-offs, and provide technical data to support their recommendations.
Equipment compatibility should be discussed early in the supplier evaluation process. Share your machine specifications, production speeds, and any known challenges with film performance. Experienced suppliers can identify potential issues and suggest solutions before production begins.
Minimum order quantities (MOQs) vary widely among rollstock packaging suppliers. MOQs affect inventory costs, cash flow, and flexibility. Brands should balance the cost savings of larger orders with the risk of obsolescence, especially for new products, seasonal items, or frequently updated designs.
Lead times impact your ability to launch new products, respond to demand spikes, and manage inventory. Ask about standard lead times, rush order options, and how the supplier manages capacity during peak seasons. Reliable lead times are essential for maintaining production schedules and meeting customer commitments.
Artwork support can save time and prevent costly mistakes. Suppliers that offer design assistance, prepress services, and proof reviews help brands bring products to market faster and with fewer revisions. This is especially valuable for brands without in-house packaging design expertise.
Repeat orders and reorder processes should be straightforward and efficient. Once a film structure and design are finalized, reordering should require minimal lead time and coordination. Ask about how the supplier manages repeat orders, stores plates or cylinders, and handles minor design updates.
Support for growing brands is another important consideration. Nutraceutical companies often experience rapid growth, requiring packaging solutions that scale with production volumes, new product launches, and expanded distribution. A supplier that understands your growth trajectory and can adapt to changing needs is a valuable long-term partner.
At Western Packaging, we help companies share their products with the world by making flexible packaging truly flexible. Our team works closely with nutraceutical brands to develop custom printed rollstock, engineered film structures, and packaging solutions built around your product specifications, equipment requirements, and brand objectives.
Conclusion
Flexible rollstock packaging has become the standard for nutraceutical brands seeking high-barrier protection, production efficiency, custom branding, and format versatility. As the supplement industry continues to evolve in 2026, rollstock packaging suppliers that combine technical expertise, reliable service, and flexible solutions will be essential partners for brands navigating complex product requirements, regulatory demands, and competitive markets.
Need flexible rollstock packaging for a nutraceutical product? Talk with Western Packaging about custom printed rollstock, film structures, and packaging options built around your product and production needs.
Frequently Asked Questions
**What is the difference between rollstock and pre-made pouches?** Rollstock is supplied on rolls and used on automated form-fill-seal equipment to create packages during the filling process. Pre-made pouches are manufactured separately and delivered ready to fill. Rollstock is typically more cost-effective for high-volume production and offers better integration with automated filling lines.
**What barrier properties are most important for nutraceutical packaging?** Moisture barrier, oxygen barrier, and light barrier are the most critical for protecting supplement quality and shelf life. The specific barrier requirements depend on the product formulation, sensitivity to environmental factors, and target shelf life.
**Can flexible rollstock packaging be customized with full-color graphics?** Yes. Custom printed rollstock supports full-color graphics, gradients, photographic images, and detailed product information. Flexographic and rotogravure printing methods deliver high-quality results suitable for retail and e-commerce presentation.
**How do I know if my equipment is compatible with a specific rollstock film?** Share your machine specifications, including web width, core size, sealing temperature range, and production speed, with your rollstock packaging supplier. Experienced suppliers can evaluate compatibility and recommend films optimized for your equipment. Testing samples on your line before placing large orders is also recommended.
**Are there sustainable or recyclable rollstock options for nutraceutical packaging?** Yes. Mono-material film structures, such as all-polyethylene laminates, are designed for improved recyclability. However, not all recyclable films provide the barrier performance required for every nutraceutical product. Brands should work with their supplier to evaluate options that balance sustainability, performance, and cost.